1,3-Propane sultone
Names
Preferred IUPAC name
1,2λ6 -Oxathiolane-2,2-dione
Other names
γ-Propane sultone; 1,2-Oxathiolane, 2,2-dioxide; 3-Hydroxyl-1-propane sulfonic acid sulfone; 1-Propane sulfonic acid-3-hydroxyl-γ-sultone; Oxathiolane 2,2-dioxide
Identifiers
ChemSpider
ECHA InfoCard 100.013.017
UNII
InChI=1S/C3H6O3S/c4-7(5)3-1-2-6-7/h1-3H2
Key: FSSPGSAQUIYDCN-UHFFFAOYSA-N
InChI=1/C3H6O3S/c4-7(5)3-1-2-6-7/h1-3H2
Key: FSSPGSAQUIYDCN-UHFFFAOYAH
Properties
C 3 H 6 O 3 S
Molar mass
−1
Appearance
White crystalline solid; colorless liquid above 31 °C
Density
1.392 g/cm3 at 40 °C
Melting point
31 °C (88 °F; 304 K)
Boiling point
112 °C (234 °F; 385 K) at 1.4 mm Hg
10% (20°C)[1]
Hazards
Flash point
158 °C (316 °F; 431 K)
NIOSH
none[1]
Ca[1]
Ca [N.D.][1]
Safety data sheet (SDS)
NIH.gov
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
1,3-Propane sultone is the organosulfur compound with the formula (CH2 )3 SO3 . It is a cyclic sulfonate ester, a class of compounds called sultones .[2] [3]
Synthesis It may be prepared by the acid catalyzed reaction of allyl alcohol and sodium bisulfite .
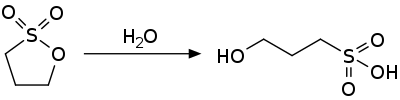
Reactions 1,3-propane sultone is an activated ester and is susceptible to nucleophilic attack. It hydrolyzes to the 3-hydroxypropylsulfonic acid.
It has been used in the synthesis of specialist surfactants , such as CHAPS detergent .[4]
Safety Typical of activated esters, 1,3-propane sultone is an alkylating agent . 1,3-Propane sultone is toxic, carcinogenic, mutagenic, and teratogenic.[5] [6]
See also References