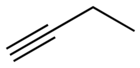
1-Butyne
H
− − -->
C
≡ ≡ -->
C
− − -->
C
|
H
H
|
− − -->
C
|
H
H
|
− − -->
H
{\displaystyle {\ce {H-C#C}}{-}{\ce {\overset {\displaystyle {H} \atop |}{\underset {| \atop \displaystyle {H}}{C}}}}{-}{\ce {\overset {\displaystyle {H} \atop |}{\underset {| \atop \displaystyle {H}}{C}}}}{\ce {-H}}}
Names
Preferred IUPAC name
Other names
Ethylacetylene
Identifiers
ChEBI
ChemSpider
ECHA InfoCard 100.003.139
EC Number
UNII
UN number
2452
InChI=1S/C4H6/c1-3-4-2/h1H,4H2,2H3
Y Key: KDKYADYSIPSCCQ-UHFFFAOYSA-N
Y InChI=1/C4H6/c1-3-4-2/h1H,4H2,2H3
Key: KDKYADYSIPSCCQ-UHFFFAOYAI
Properties[1]
C4 H6
Molar mass
54.091 g/mol
Density
0.6783 g cm−3 [1]
Melting point
−125.7 °C (−194.3 °F; 147.5 K)[1]
Boiling point
8.08 °C (46.54 °F; 281.23 K)[1]
Hazards
GHS labelling
Danger
H220 , H280
P210 , P377 , P381 , P403
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
1-Butyne is an organic compound with the formula CH3 CH2 C≡CH . It is a terminal alkyne . The compound is a common terminal alkyne substrate in diverse studies of catalysis. It is a colorless combustible gas .[1]
1-Butyne participates in reactions typical for terminal alkynes, such as alkyne metathesis ,[2] formaldehyde . Based on its heat of combustion , it is slightly more stable than its isomer 2-butyne .[3]
See also References
^ a b c d e Lide, David R. (2008). CRC Handbook of Chemistry and Physics, 89th Edition . CRC Press . pp. 3–84. ISBN 978-0-8493-0488-0 ^ Zhang, Wei; Kraft, Stefan; Moore, Jeffrey S. (2004). "Highly Active Trialkoxymolybdenum(VI) Alkylidyne Catalysts Synthesized by a Reductive Recycle Strategy". Journal of the American Chemical Society . 126 (1): 329–335. doi :10.1021/ja0379868 . PMID 14709099 . ^ Prosen, E.J.; Maron, F.W.; Rossini, F.D. (1951). "Heats of combustion, formation, and insomerization of ten C4 hydrocarbons" . Journal of Research of the National Bureau of Standards . 46 (2): 106. doi :10.6028/jres.046.015
External links